 Calculating pI values įor an amino acid with only one amine and one carboxyl group, the pI can be calculated from the mean of the pKas of this molecule. When the environment is at a pH value equal to the protein's pI, the net charge is zero, and the protein is not bound to any exchanger, and therefore, can be eluted out. At high pH values, the net charge of most proteins is negative, where they bind to the positively-charged matrix in anion exchangers. At low pH values, the net charge of most proteins in the mixture is positive - in cation exchangers, these positively-charged proteins bind to the negatively-charged matrix. When a mixture containing a target protein is loaded into an ion exchanger, the stationary matrix can be either positively-charged (for mobile anions) or negatively-charged (for mobile cations). Buffers of various pH can be used for this purification process to change the pH of the environment. The specific pI of the target protein can be used to model the process around and the compound can then be purified from the rest of the mixture. Biological proteins are made up of zwitterionic amino acid compounds the net charge of these proteins can be positive or negative depending on the pH of the environment. In biomolecules, proteins can be separated by ion exchange chromatography. Isoelectric focusing is also the first step in 2-D gel polyacrylamide gel electrophoresis. Proteins can, thus, be separated by net charge in a polyacrylamide gel using either preparative gel electrophoresis, which uses a constant pH to separate proteins or isoelectric focusing, which uses a pH gradient to separate proteins. At a pH below their pI, proteins carry a net positive charge above their pI they carry a net negative charge. Amino acids that make up proteins may be positive, negative, neutral, or polar in nature, and together give a protein its overall charge. Biological amphoteric molecules such as proteins contain both acidic and basic functional groups. Such molecules have minimum solubility in water or salt solutions at the pH that corresponds to their pI and often precipitate out of solution. The pI value can affect the solubility of a molecule at a given pH. In the common case when the surface charge-determining ions are H +/HO −, the net surface charge is affected by the pH of the liquid in which the solid is submerged. Surfaces naturally charge to form a double layer. 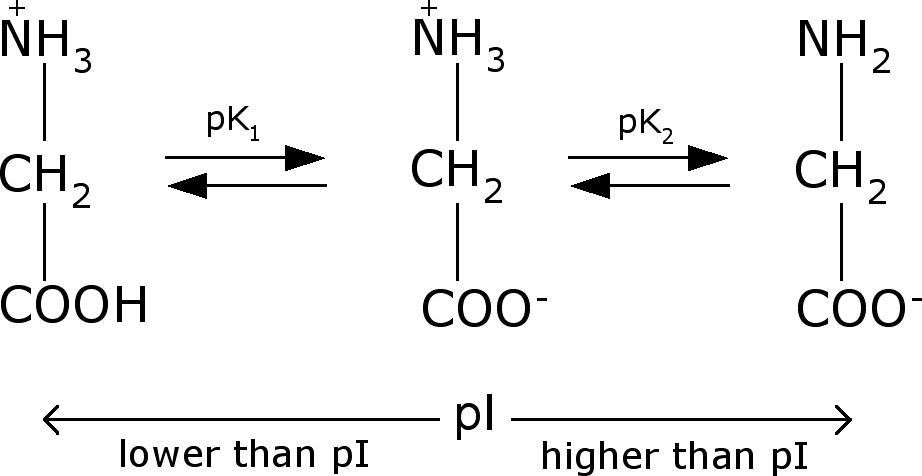 
The net charge on the molecule is affected by pH of its surrounding environment and can become more positively or negatively charged due to the gain or loss, respectively, of protons (H +). The standard nomenclature to represent the isoelectric point is pH(I). 
The isoelectric point ( pI, pH(I), IEP), is the pH at which a molecule carries no net electrical charge or is electrically neutral in the statistical mean. PH at which a particular molecule, or the surface of a given solid, carries no net electrical charge 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
